The quality of air within an IVF lab will inevitably affect embryonic development and therefore has a critical impact on IVF success. Our guest blogger Giles Palmer is a senior clinical embryologist who has supported many IVF labs in improving the air quality. In this blog post he shares his valuable recommendations on how to secure the air quality in your IVF lab.
Hazardous components in air can come from many sources
Air in urban areas can contain high levels of pollutants such as;
- carbon monoxide
- nitrous oxide
- sulphur dioxide
- heavy metals.
Likewise, indoors, construction materials, MDF (medium-density fiberboard), PVC flooring, paints and adhesives constitute the major source of volatile organic compounds (VOCs), which can also contribute to the phenomenon called “sick building syndrome”1. Other sources of indoor chemical hazards are cleaning fluids, floor waxes, cosmetics and cigarette smoke.
National Health and Safety Authorities recommend safe limits of VOC exposure for humans and give guidelines for building ventilation, but there is little evidence in the IVF literature of the toxicological effect from these substances on embryos in vitro.
Controlling air quality in the IVF lab has beneficial effect on results
Pollutants can settle on work surfaces and dissolve in aqueous solutions of embryo culture medium and even more so in the lipophilic oil overlays. The vulnerable embryos cannot protect themselves against these environmental contaminants.
A study has shown that not only are high levels of aldehydes and other noxious compounds present in the IVF laboratory (higher than outside air and the average house) but also in the incubators2.
Controlling air quality in an IVF laboratory has shown beneficial effects regarding fertilisation and embryo development3.
The EU directive 2004/23/EC stipulates air quality requirements when human tissue and cells are handled. A critical point is clean air, which has let to centres making - at the very least - slight structural changes to their IVF units.
Location important factor in reducing air pollution
One of the most important design considerations should be concerning location, especially regarding the positions of the laboratory air intakes, the location of the lab within the building and even the location of the building itself. These considerations and challenges for reducing air pollution must factor into the initial design phase of the lab.
Since most centres are located in cities and towns, control of the air quality may be hindered in the urban air, especially for units close to busy roads and car parks. Within a hospital, difficulties may be encountered with working in an area adjacent to the laundry, sterilizing or histology departments.
A clean room environment secures better outcome
If embryo development and implantation depends so highly on its culture environment, the IVF laboratory should do well to apply more stringent regulatory standards in matters of particle control and especially molecular air quality. Only then will the embryology laboratory truly enter the biotechnology arena.
“Clean room technology” creates a carefully controlled sealed environment in which the entry of particles and contaminants is significantly minimised. This is achieved by highly filtered air being flushed through High Efficiency Particle Air (HEPA) filters under positive pressure, which leads to a cleaner environment overall. New to the IVF industry are filters housed within the ducts that use chemical/absorbant filtration and/or UV light irradiation, photocatalytic conversion or a combination of some or all of the above methods.
All furniture, cleaning methods and clothing should be clean room approved. The laboratory should have smooth, non-porous walls, impervious unbroken surfaces with corners and ledges that are easy to clean, no sliding doors, sinks and drains. The lighting should be within sealed lighting units in order to help reduce airborne particles.
Cladding with a non-porous material such as panels made from aluminium tri-hydrate are expensive but provide an inert (low VOC), hypoallergenic, easy to clean wall.
Ducts and pipes can be insulated internally - i.e. hidden between wall panels - or externally (covered within an alcove) to prevent dust accumulation.
If designing from scratch, hollow modular walls that fit together like pieces of “lego” can be assembled to encase an area of interest; providing an enclosed controlled environment with hidden ducts and power supplies meeting all the needs of a modern IVF laboratory. Stand-alone prefabricated laboratory constructs called Clean Room Cells (CRC) that can be easy assembled on site which offer an economical solution to obtaining an instant cleanroom environment within pre-existing buildings – or in locations with poor overall air quality - or in the absence of local materials and expertise.
Traditionally copper pipes have been used to transport medical grade gasses from cylinder to incubator. Copper, however is prone to oxidation and therefore inert stainless steel tubing certified for the use with medical gasses is now recommended despite the scarcity of clinical evidence supporting its use.
Don’t forget the rooms adjacent to the lab
Oocyte retrieval and embryo transfer which demands cooperation with the adjacent operating theatre creates a challenge for maintaining clean air conditions, therefore, hermetically sealed doors and pass through windows may help these critical steps.
Changing rooms should be as clean as possible and adjacent to the laboratory. Positive pressure in the laboratory should be extended to the adjoining operating theatre which also has sealed doors and ceilings.
Provision should be given to how the bulky IVF lab equipment will enter the unit after the construction phase. A removable sealed section of wall allows not only ventilation (VOC release) but an easy way to bring large equipment such as incubators and laminar flow cabinets during construction and during the “off gassing stage”. Provisions should be made so that doors are wide enough to accommodate large equipment after the lab becomes permanently sealed and operational.
Allow new equipment to be “run in”
When at all possible new incubators should be “run in” in a room other than the laboratory for a certain time period to dissipate latent VOCs produced in their manufacture. This may make the difference between a poor initial pregnancy rate or triumphant start to a new IVF unit!
How can an existing laboratory improve its air quality?
Other than installing a central air handling system that includes activated carbon pre-filters, chemical filters, photo catalytic conversion and HEPA filters, several commercial mobile filter units exist which are somewhat efficient (despite lacking some of the more modern air purification technologies) by purifying the air of damaging volatile compounds, bacteria and moulds4. Air is forced through the device and either passed through carbon filters, ultra violet light for photocatalytic purification and/or chemical filters.
Compressed gas can contain harmful compounds such as benzene, isopropanol and pentane2. Medical grade N2 and CO2 can be filtered to remove any lingering contaminants before entering the incubators by gas line filters.
As high levels of VOCs have been shown to be present incubators, internal air filtering systems may be added to decrease VOC levels inside incubators themselves5,6.
Also since anaesthetic gasses use in oocytes retrieval may linger in the air a suitable extraction system may be installed in the operation theatre
To reduce VOC emissions, wooden laboratory shelves, cupboards and tables should be replaced with stainless furniture and workbenches used in pharmaceutical electronic and food industry.
Keep restricted access
Unauthorized personnel, packaging and other materials into the laboratory should be restricted. Installing an air lock chamber before entering the laboratory while expensive and primarily aimed at reducing particle contamination offers both a physical barrier and acts as an “air shower” when flushed with filtered air.
Staff can be one of the biggest contaminants in a clean environment. General good hygiene must be practiced. Exhaled air from smokers contains residual tobacco particles (0.5 and 2 micron in size) that can remain indefinitely in the air or settle and on surfaces and walls. Therefore clean-room etiquette should exclude smokers or at least discourage them from smoking within the working day.
Laboratory dress code
Clean room clothing provide maximum control against microbial and particle contamination. Cotton hospital “greens” should be replaced by pocket-less trouser suits or two piece suits with covered wrists and neck. Made specifically for clean rooms these polyester garments act as a particle barriers which shred no fibres into the environment. In addition, special treatment of the garments during laundering should always be aimed at reducing residual traces of laundry detergent.
Be careful when renovating
Renovation of an existing laboratory can introduce a variety of compounds into the work place and emission of harmful compounds used in finishing can have an adverse effect on embryo development and pregnancy rate either temporarily or long term!
Traditionally, paints, adhesives and sealants can contain alkanes, aromatics, alcohols, aldehydes and ketones amongst others. Epoxy paints emit VOCs and take several weeks to cure and thus their use should be avoided.
When renovating an older laboratory, there are materials that can be used with less harmful chemicals: solvent free adhesives used for floor covering are available with low VOC emissions as well as “ecological” water based paints that contain non-volatile chemicals.
On site supervision is essential to assure no materials or methods are used that could jeopardize later IVF success.
Results from our renovated laboratory
Sufficient time to purge the room of harmful chemicals should be allocated before laboratory start up. At least for one week room temperature and positive air pressure should be increased dramatically aiding removal of VOCs introduced in the construction processes.
Following the above measures we renovated our laboratory in Athens, Greece. The laboratory premises were already fitted with Activated Carbon pre filters and HEPA and in-line filters but an upgrade to a Clean room GMP Class C (ISO class 7) was sanctified. Upgrades included a complete refurbishment of the lab and its layout, installation of Corian® panels for the walls, installation of new filtered ducts and of an airlock at the entrance of the lab.
Completion took one month - during which the laboratory was inactive - and VOC measurements of the environment using ACS badges were measured during each critical phase of reconstruction and two weeks following.
Highest VOC levels were observed in the adhesion of Corian © wall panels where acetone reached a level of 15.5 ppm. Vinyl Glue used in the final stages of floor surfacing also indicated emissions of hazardous VOCs. One week of purging was performed after completion. Two weeks after completion no detectable chemicals were present.
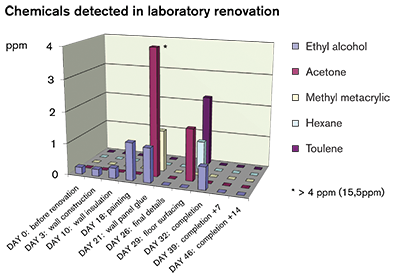
Figure 1 illustrates the VOC emissions in various phases before and after renovation
It is up to every IVF facility to access the clean air requirements and determine if changes need to be made.
Maintaining good air quality through good laboratory practice
To maintain the clean air environment good laboratory practice must be continued. Frequent recording of air flow parameters (specifically of filter integrity and daily positive pressure measurements) and servicing of equipment safeguards against drop in air quality due to exhausted HEPA filters.
Positive pressure between the laboratory and adjacent rooms can be monitored by the pressure differential recorded daily via a simple pressure gauge to indicate filter efficiency.
Validation of air quality can be measured periodically by particle counters. To measure workplace exposure of toxic vapours - VOC levels can be monitored either electronically or inexpensively with organic chemical sensors such as ACS badges that analyse absorbed ion levels on chromatography paper. ACS badges are an inexpensive and simple method for monitoring VOC levels and can be used for quick and regular measurements, but it should be noted that they have a low resolution when compared to other more sensitive methods.

Swab test can determine microbial count
One further measure that can detect contamination from the operator (i.e. from sweat or lapses in hygiene protocols) and should be introduced to your total quality management system is tactical swab tests of all work stations and critical areas to determine microbial count. Agar contact plates or commercially available kits count microbial load on lab surfaces. The practice of using key performance indicators such as in vitro fertilization rate, cleavage rates, embryo quality and pregnancy rates adds to your total quality management.
References
- Takigawa T,Wang BL, Saijo Y, Morimoto K, Nakayama K, Tanaka M, Shibata E, Yoshimura T, Chikara H, Ogino K, Kishi R. Relationship between indoor chemical concentrations and subjective symptoms associated with sick building syndrome in newly built houses in Japan. Int Arch Occup Environ Health. 2010 Feb;83(2):225-35.
- Cohen, A. Gilligan, W. Esposito, T. Schimmel, B. Dale (1997). Ambient air and its potential effects on conception in vitro. Hum. Reprod. Vol 12 (8) pp 1742-1749.
- R Boone, J.E. Johnson, A. Locke, M.M. Crane, T.M. Price (1997). Control of air quality in an assisted reproductive technology laboratory. Fertil. Steril. Vol 71 (1) pp150-154.
- Forman, V. Polanski, A. Gilligan, D. Reiger. (2004) Reduction in volatile organic compounds, adehydes, and particulate air contaminants in an IVF laboratory by centralized and standalone air filtration systems. Fertil Steril Vol. 82, Suppl.2. P-535 (Abstract).
- Hall, A. Gilligan, T. Scchimmel, M. Cecchi, J. Cohen (1998). The origin, effects and control of air pollution in laboratories for human embryo culture. Hum. Reprod. Vol 13 (4) pp 146-155.
- J.F.Mayer, F. Nehchiri, V.M Weedon et al . (1999) Prospective randomized crossover analysis of the impact of an IVF incubator air filtration system (Coda. Gen X) on Clinical pregnancy rates. Fertil Steril Vol. 72, Suppl. 1. S42.
The opinions expressed by Vitrolife’s guest bloggers are their own and do not necessarily reflect Vitrolife's opinions or views.
Topics: IVF laboratory control, Embryo culture & transfer
Written by Giles Palmer
Giles Palmer is the Director of the Embryology Department of the Assisted Reproduction Unit at MITERA hospital in Athens and an ESHRE certified senior clinical embryologist. His collaboration with Agia Sophia Children's Hospital (Athens University) has led to many publications in leading scientific journals in the field of reproduction. Apart from working at Mitera, he is also supporting other labs in QC-related matters.
