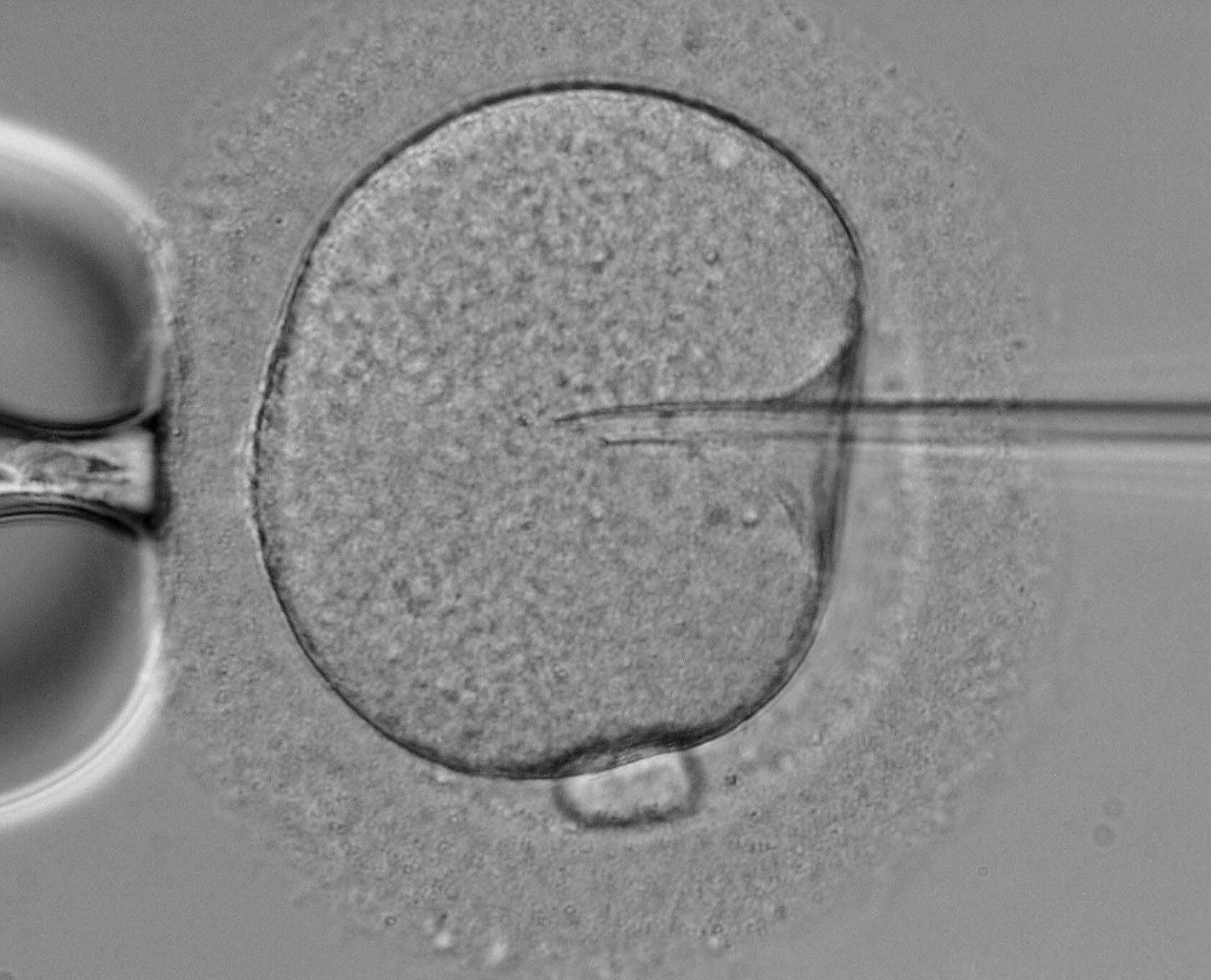
In the intricate world of assisted reproductive technology (ART), every minute detail can significantly influence the outcome. One such detail that has garnered increasing attention is the choice of buffer during Intracytoplasmic Sperm Injection (ICSI) in the in vitro fertilization (IVF) laboratory. The meticulous research conducted by Dr. Robert J. Mendola, a distinguished expert in clinical IVF embryology, sheds light on the profound implications of different buffers on embryo development. Learn more about Dr. Robert J. Medolas findings in this blog post.
How different buffers during ICSI can impact the development of embryos in the IVF laboratory
By Robert J. Mendola, Ph.D., HCLD, May 23, 2024
Comparison of closed vs open vitrification devices
By Vitrolife Group, Jan 27, 2022
The concept of fast freezing or vitrification was first described more than 80 years ago by Basile J. Luyet, the so-called Father of Cryobiology (Luyet, 1937). He showed that supercooled solutions could be solidified without crystallization, forming a glass-like state. Already then, the potential of the technique and the associated challenges were in the research spotlight. Today, we’ve managed to overcome all methodological-related issues of vitrification. It has evolved into a reliable and efficient method to freeze oocytes and embryos. Vitrification is used for medically assisted reproduction and fertility preservation: the goal is to ensure the maximum survival rate with the highest level of biosafety. In this blog post, we will compare closed and open carrier devices for vitrification.
Can a culture oil improve embryo development?
By Dmitry Nikiforov, Dec 16, 2021
The simple answer is, it can’t. In a well-functioning culture system, the oil should only act as a cover, protecting the gametes and embryos from changes in the environment and potential contaminants. But if the oil quality is sub-optimal, it can decrease embryo development. Most oils are produced from petroleum, which means that embryotoxic components may be present due to the production process and origin of the raw materials.
This year, the annual meeting of ASRM 2021 was back in its physical form, and we where there in-person with a great team from Vitrolife. For those of you who couldn´t attend the meeting or want to update yourself again on the interesting sessions, we have compiled this blog post, with thoughts and reflections from the Vitrolife team on some of the scientific content presented at the meeting.
Vitrolife´s integrated approach to sustainability
By Rikard Ledin da Rosa, Oct 15, 2021
Why there is a need for the sustainable development goals
The establishment in 2015 of the Sustainable Development Goals (SDGs) has created a global consensus on the development framework for a better world. However, as we look to the next decade, there is significant cause for concern about the future. The IPCC’s Sixth Assessment report, released in August 2021, confirms that it is indisputable that human influence has warmed the climate system, raising global surface temperature. The report confirms that there is no going back from some changes that are already affecting the climate system. These changes in the climate are widespread, rapid and intensifying and impacts are affecting every region on Earth, including the oceans.
How Vitrolife prepares for the new European Medical Device Regulations
By Hans Lehmann, Sep 22, 2021
Introduction to Medical Device Regulations - MDR
For 30 years medical devices have been governed by the Medical Device Directive (MDD) implemented in EU/EEA in 1993, and there have been quite a few developments in the field of medical devices since then. For instance, apps used as stand-alone products for different medical purposes have become a reality. The general population has become older, patients are more used to finding medical information on the internet, and the beauty industry uses more advanced and invasive techniques than before. These changes, and more, have prompted an update of the control of medical devices for some time. Eventually, the new Medical Device Regulations (MDR) were developed and came into force on May 26, 2021. From this date, all new medical devices and the manufacturer’s quality management system must comply with the MDR. Implementation deadlines for existing devices are dependent on classification and expiry of existing MDD certificates.
Thoughts and reflections from ASRM 2020
By Vitrolife Group, Nov 10, 2020
This year, online was the place to be when participating in the 76th annual meeting of ASRM. In this blog post, thoughts and reflections from the Vitrolife team have been collected on some of the scientific content presented at the meeting.
Thoughts and reflections from ESHRE 2020
By Vitrolife Group, Aug 20, 2020
The 36th annual meeting of ESHRE took place online this year, with more than 12,000 registered participants. Like last year, the Vitrolife team has put together our thoughts and reflections on some of the scientific content from the meeting. We also offer the opportunity to watch our recorded scientific symposium again.
Memoriam for Prof Michelle Lane
By Dr. Mark Larman, Feb 12, 2020
It is with great sadness that Vitrolife posts this memoriam for Prof Michelle Lane, who passed away last week.
The value of a true partnership – a story from Aleris-Hamlet Fertility, Copenhagen, Denmark
By Vitrolife Group, Jan 15, 2020
What do you as an IVF professional look for when setting up an entirely new clinic and laboratory? For the laboratory manager, Helle Bendtsen at Aleris-Hamlet Fertility in Copenhagen, the advantage of having one supplier for all of their procedures in the laboratory was clear quite quickly: ‘When setting up the clinic we received great support from Vitrolife in terms of selecting all the products for the entire IVF procedure. From oocyte retrieval all the way to vitrification and transfer’, she says and adds: ‘Previously I’ve worked with products from several different suppliers, but now I can truly see the benefits with having all our products from Vitrolife.’