Laser systems have been an integral part of routine IVF laboratory work for many years. Developed by Dr Klaus Rink and taken to first clinical application in 1994 in collaboration with Dr Alfred Senn, the Octax laser systems quickly found worldwide adoption. Applications of Octax laser systems include assisted hatching / assisted thinning, blastocyst collapsing, trophectoderm and blastomere biopsy and last but not least sperm viability testing. In 2000 Dr Markus Montag was the first author who described laser-induced immobilisation of human sperm (1). In 2004 he further specified this application by describing the method of laser-assisted viability testing (2).
Sperm viability testing using Octax Laser
A prerequisite for initiating fertilization is, of course, the use of viable sperm. Only a viable spermatozoon will induce correct oocyte activation and subsequent viable embryo development. For ICSI procedures usually a
sperm´s motility is the most obvious sign of viability. However, in some TESE cases or cases of complete asthenozoospermia, e.g., flagellum defects, it is impossible to select viability via motility. Non-motility of viable sperm can be caused by different reasons: The first reason is a genetic defect which results in the structural inability of all spermatozoa to move their flagellum. The second reason for immobility is heterogenous events, like oxidative stress, infections, testicular samples from obstructive or non-obstructive reasons and several other influences.
In our daily lab routine, the embryologists often must face problems with highly impaired spermatozoan quality and even worse, sometimes we only have immotile spermatozoa for ICSI. There is the potential possibility to randomly select immotile spermatozoa, which can be viable or non-viable for ICSI, which will result in very low fertilization rates. The use of only viable spermatozoa is of course preferable and increases the fertilization rates.
Therefore, it is important to use routine methods, which can differentiate between immotile dead and immotile viable spermatozoa. Well-known methods are: the hypo-osmotic swelling test (HOST), sperm selection with activating substances, sperm tail flexibility test and last but not least the laser assisted immotile sperm selection (3).
In cases of immotile spermatozoa with TESE cases or severe asthenozoospermia we have very good and promising results with the laser assisted sperm selection. We prioritise this method over the others, because of its quick, easy, and reliable application. Artificial activation with cell-permeable substances will not work with patients with flagellum-defects. Furthermore, it's possible intracellular downstream effects make this method not the first choice for us.
Of course, the ICSI microscope must be equipped with a laser, which is nowadays a rather standard piece of equipment due to other routine laser applications. The use of a laser for sperm viability testing is very reliable and does not depend on extensive result interpretations. The use of the laser viability testing method is not difficult to learn, but still requires careful training and some experience from the embryologist.
For sperm laser treatment, the first step is to set up an ICSI dish. Several droplets of 10 - 20 µl MOPS and / or HEPES buffered medium are set up under oil to which prepared sperm will be added.
In the second step we incubate 1 -2 µl of washed and prepared spermatozoa into the droplets of the prewarmed dish provided for the sperm. The sperm concentration should not be too high, so that you have enough space around each spermatozoon to apply laser pulses to one single sperm cell. We look for a spermatozoon, with a relatively good morphology (if you have the chance to do so). Then we arm our Octax laser and set it to a pulse time of 4-5 ms with laser setting: “hole”. One laser pulse is applied to the end of the tail, as far away from the sperm-head as possible. Now it is very important to watch the tail reaction very accurate and not to lose the tail out of sight while releasing the laser pulse. There are several possible reactions: curling or shrinking of the tail and a little jerking of the sperm is also possible and a very common reaction (see Fig. 1). We describe these sperm cells as presumably alive, transfer them to PVP and use them for ICSI within 10 min after laser treatment. Spermatozoa that do not respond to the laser pulse are regarded as dead and are not used for ICSI. We can use the static or movable Octax laser for performing this method. Of course, using the movable Octax Navilase is a more time-efficient procedure for us.
Figure 1: The road to laser-assisted viability testing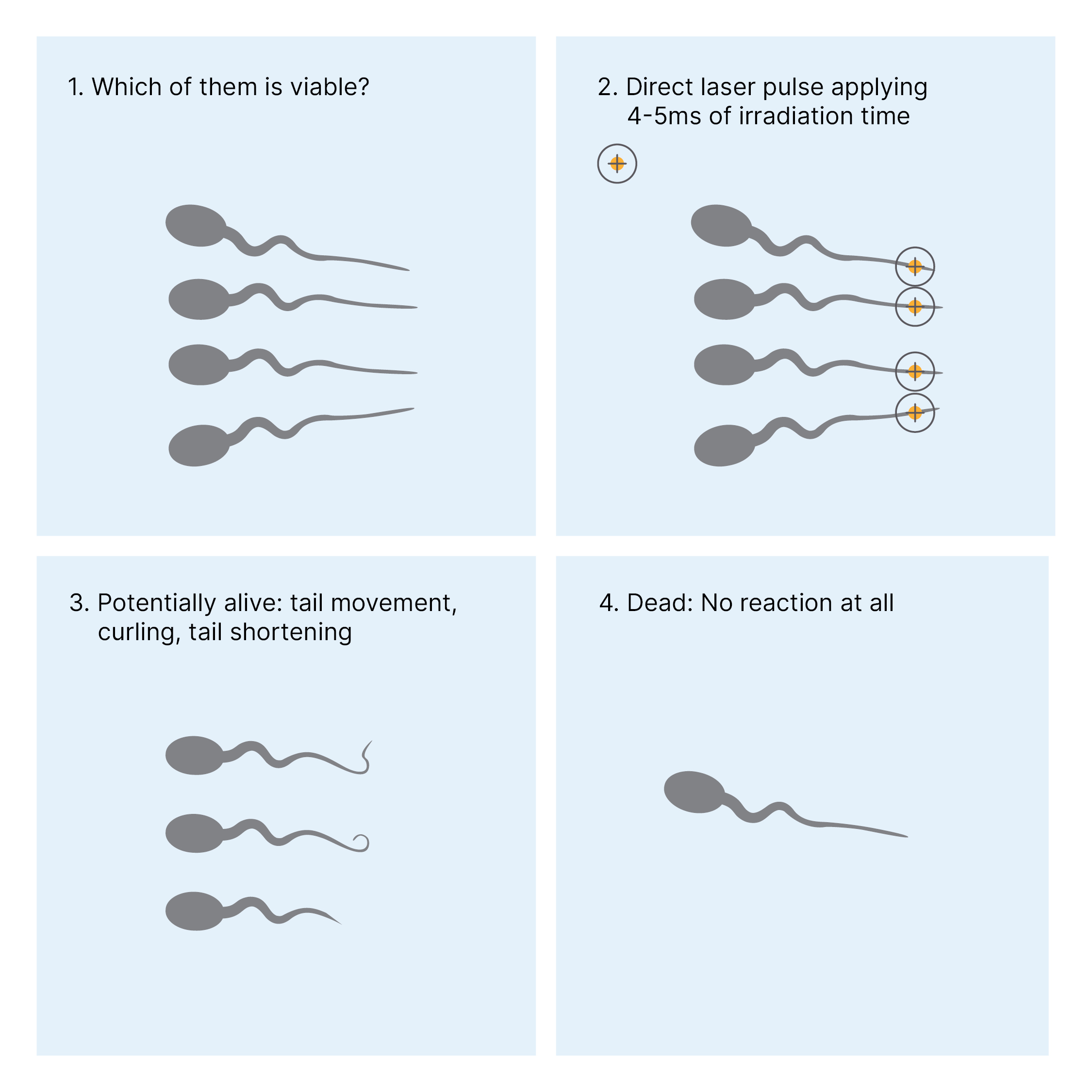
We still have no direct proof that those spermatozoa which show one of the above reactions to the laser pulse are indeed viable. However, in validation experiments we assessed sperm viability via the eosin live / dead test and found out that the percentage of presumably viable spermatozoa correspond to the eosin test results. Furthermore, we have relatively high fertilization rates (up to 90%), which support our classification theory. The fact that several live born babies would have not been born without this method, support us to use this method as the first choice in severe cases of asthenozoospermia.
Based on the regulatory status not all applications are approved in all countries. Please contact your local sales representative on status in your country.
References:
- Montag M, Rink K, Delacretaz G, van der Ven H. Laser-induced immobilization and plasma membrane permeabilization in human spermatozoa. Hum Reprod. 2000;15(4):846-52.
- Aktan TM, Montag M, Duman S, Gorkemli H, Rink K, Yurdakul T. Use of a laser to detect viable but immotile spermatozoa. Andrologia. 2004;36(6):366-9.
- Nordhoff V. How to select immotile but viable spermatozoa on the day of intracytoplasmic sperm injection? An embryologist's view. Andrology. 2015;3(2):156-62.
Written by Dr Claudia Staib
Dr Claudia Staib has a PhD in Microbiology and is working in the field of reproductive biology since 2007. She is senior embryologist (ESHRE) and the lab director in the ZERM (Center for Gynaecologic Endocrinology and Reproductive Medicine) at the Hospital for Gynaecology and Obstetrics in Wuerzburg, Germany. Recently, the ZERM has opened up a new laboratory where state of the art technology, quality issues and workflow efficiency are working hand in hand.

