Preimplantation Genetic Screening (PGS) is a procedure designed to examine embryos for chromosomal abnormalities with the aim of de-selecting embryos with low implantation potential. Preimplantation genetic diagnosis (PGD) refers to genetic diagnosis of embryos prior to transfer in cases with a known inherited disorder. Vitrolife has conducted a survey within the IVF community to determine how wide spread the use of these procedures actually is in the everyday routines. The results will be shared in this blog post.
Participant demographics
Survey participants came from all around the globe, with most representatives from Europe and North America. The majority of respondents work in a private IVF-clinic and 87% of them are embryologists and laboratory managers.
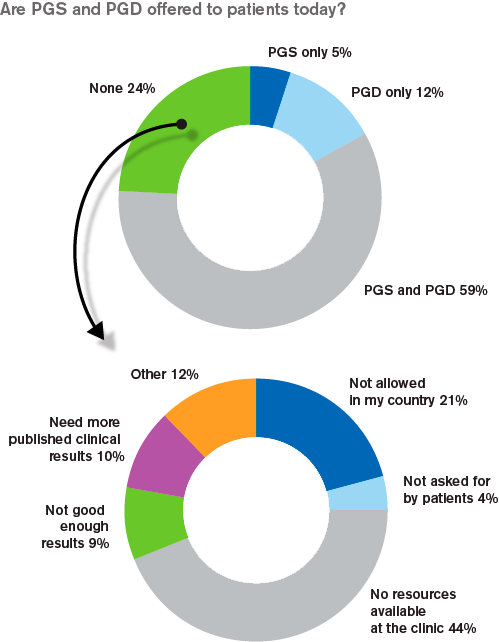
Are PGS and PGD offered to patients today?
The majority of the responding clinics (59%) offer both PGS and PGD, whereas almost a quarter (24%) does not offer any of them. The reasons for not offering PGS are mainly either legal or due to lacking resources available at the clinic to perform the procedures.
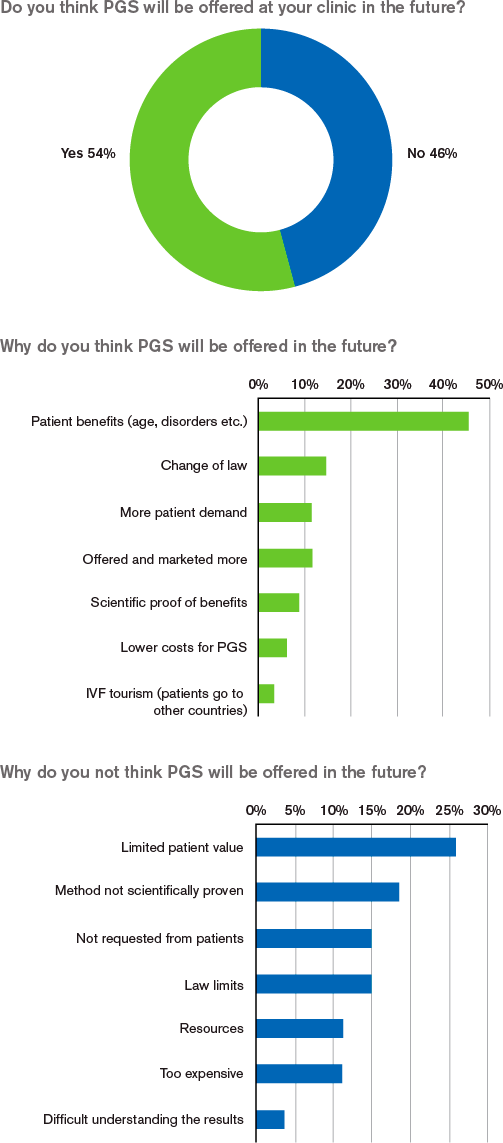
Patient demands have major impact on the clinical practice
Whether a clinic chooses to offer PGS/PGD or not appears to be mirroring the same factors: Patient value and benefits, patient requests, legal status and scientific proof. The patient demands, in particular, have a major impact on the clinical practice. PGS must be perceived as a value by patients, it must be legally allowed and actively requested.
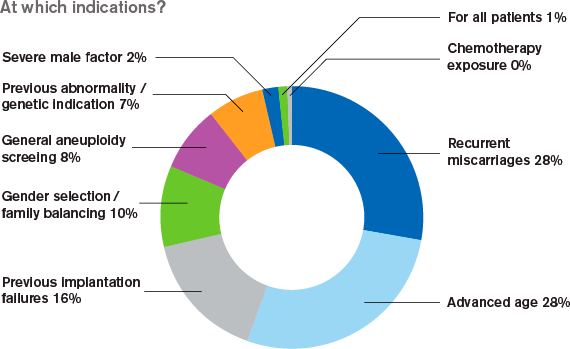
For which indications is PGS used clinically?
The most common indications for using PGS clinically are recurrent miscarriages, advanced maternal age (AMA) and previous implantation failures. Performing PGS is not only due to high patient age, but also to previously failed treatments. The possibility to offer genetic screening for couples with failed treatments will be a pull factor in competition between clinics. Regulation on IVF treatment differs between countries and services offered can be available more in private or in public IVF clinics. This will result in patients seeking certain treatment in different clinics or different countries as one of the last options.
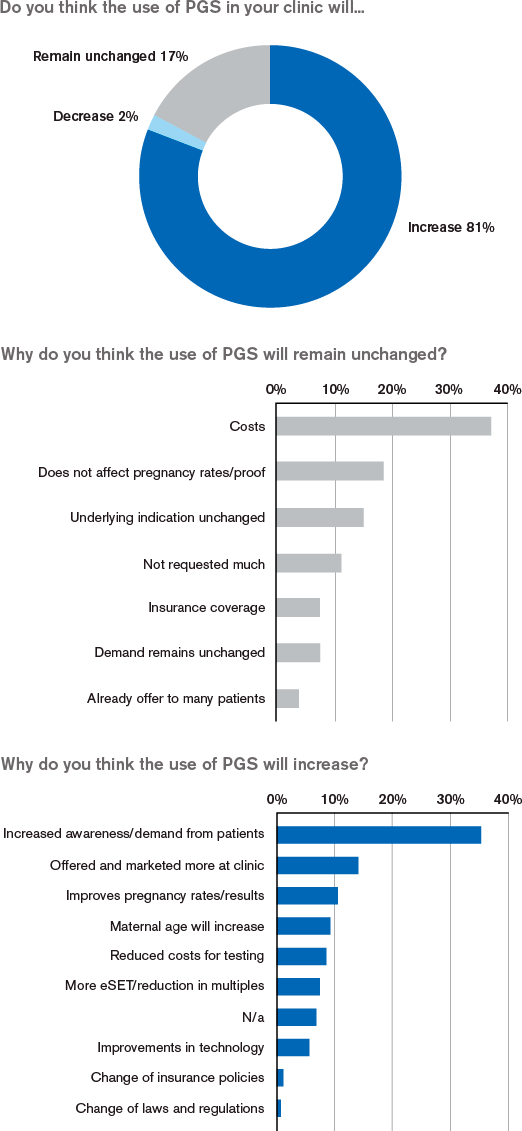
The use of PGS will increase in the future
Currently, according to this survey, less than 5% of IVF patients have PGS included in their treatment. However, 81% of the respondents believe that the use of PGS will increase in the future, mainly because of an increased awareness and demand for the service. The active marketing of the tests is also mentioned as a reason for increased use of PGS. Finally, improvement of pregnancy rates and the fact that the maternal age will increase are seen as supporting factors for the increased use of PGS. Most common reasons mentioned why the frequency of PGS will remain unchanged are costs and that the proof of increased pregnancy rates is currently lacking.
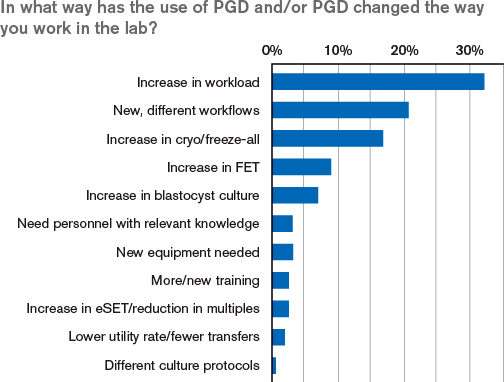
The use of PGS and/or PGD has changed the way embryologists work
The majority of the respondents point at a change in routine work due to the introduction of PGS and/or PGD in the IVF laboratory. The two main reasons are an increase in workload and new, different workflows in the laboratory. Interestingly, the following reasons are due to changes in existing procedures. Because of the fact that analysis is made on predominantly blastocysts, more blastocyst culture is expected and subsequently the method of cryopreservation is vitrification. With the use of genetic testing, an increase is seen in cryopreservation and frozen embryo transfers. These three effects are logical results of the need to cryopreserve embryos after sampling of the cells for testing and waiting for results.
Finally, knowledge, training and equipment are factors influencing the way embryologists work since PGS/PGD is not part of a basic IVF laboratory education today but need further training. Other effects include the increased need for witnessing, communication regarding results, and a new set of paperwork and logistical flows.
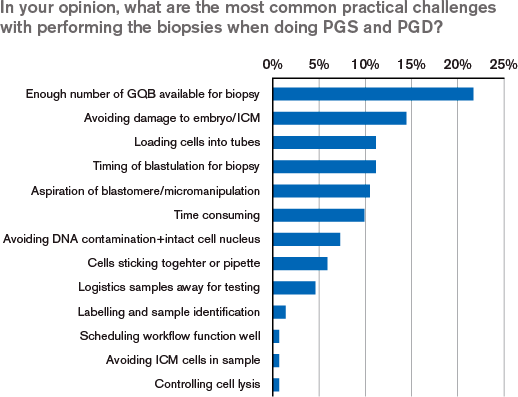
The challenges embryologists face
Embryologists face several practical challenges when performing PGS and PGD. Predominantly, blastocyst biopsies are performed, which entails having as many good quality blastocysts (GQB) as possible to biopsy. This adds another dimension to blastocyst culture. Except for the deselection of non-developing embryos, the synchronisation of blastulation now becomes even more important. Besides optimising culture conditions, such as the use of low oxygen, there are not that many possibilities to improve synchronisation of a cohort of embryos in a given patient.
Together with a high number of blastocysts at the day of sampling come also challenges on the choice of embryos that can be used successfully for biopsy. Besides checking at specific time points, the challenge for the embryologists is to decide when to perform the biopsies.
Apart from blastocyst culture and time challenges, there are more method-specific areas mentioned as challenges. For example avoiding damage to the embryo when performing the biopsy, cells sticking together, avoiding ICM cells in the sample, avoiding DNA contamination and loading cells into tubes (called “tubing”). These challenges reside in how the samples are obtained and the specific requirements a DNA test places on the procedure. A balancing dilemma is to have enough cells for analysis while minimising the damage on the embryo by removing the cells.
These are the areas that require training in both practical and theoretical terms. Logistics and labelling are mentioned as small challenges.
A “new normal” for IVF clinics
With the introduction of PGS in the IVF clinic, embryologists face having to change the workflows in the lab. In IVF procedures, the effects are increase of blastocyst culture, increase of cryopreservation (vitrification), frozen embryo transfers and also single embryo transfer. An increase in the need for training, education and technical equipment is also connected to the introduction of PGS/PGD.
The IVF clinic has indeed changed into a “new normal” on many fronts. On one hand, the laboratory copes with new workflows, change in embryo culture, micromanipulation and cryopreservation. But on the other hand, the IVF laboratory also face the challenge of managing more patients with frozen blastocyst transfers and in some clinics more single embryo transfers.
In conclusion
The main insights from this survey are that PGS is offered mainly for patients with advanced maternal age and recurrent miscarriage, mostly representing 5% or less of patients at the clinics which participated in this survey. Most respondents believe that PGS will increase in the future, driven by patient awareness/demand and more active marketing from clinics. Many of the clinics not offering PGS today are planning to start.
PGD shows similar survey results with the main indication being known monogenetic disorders, used on less than 5% of patients and the majority believes that the volume will remain stable in the future.
For both procedures, the genetic testing is frequently performed off-site and it has affected both the overall workload and workflows, while implementation of the genetic analyses methods also have increased the use of extended culture, vitrification, frozen embryo transfers and to some extent single embryo transfer.
In terms of practical challenges, having enough good quality blastocysts and timing the blastulation is a main concern, but also avoiding damage to embryos, loading of samples and aspiration technique.
Topics: genetic testing, IVF community insights
