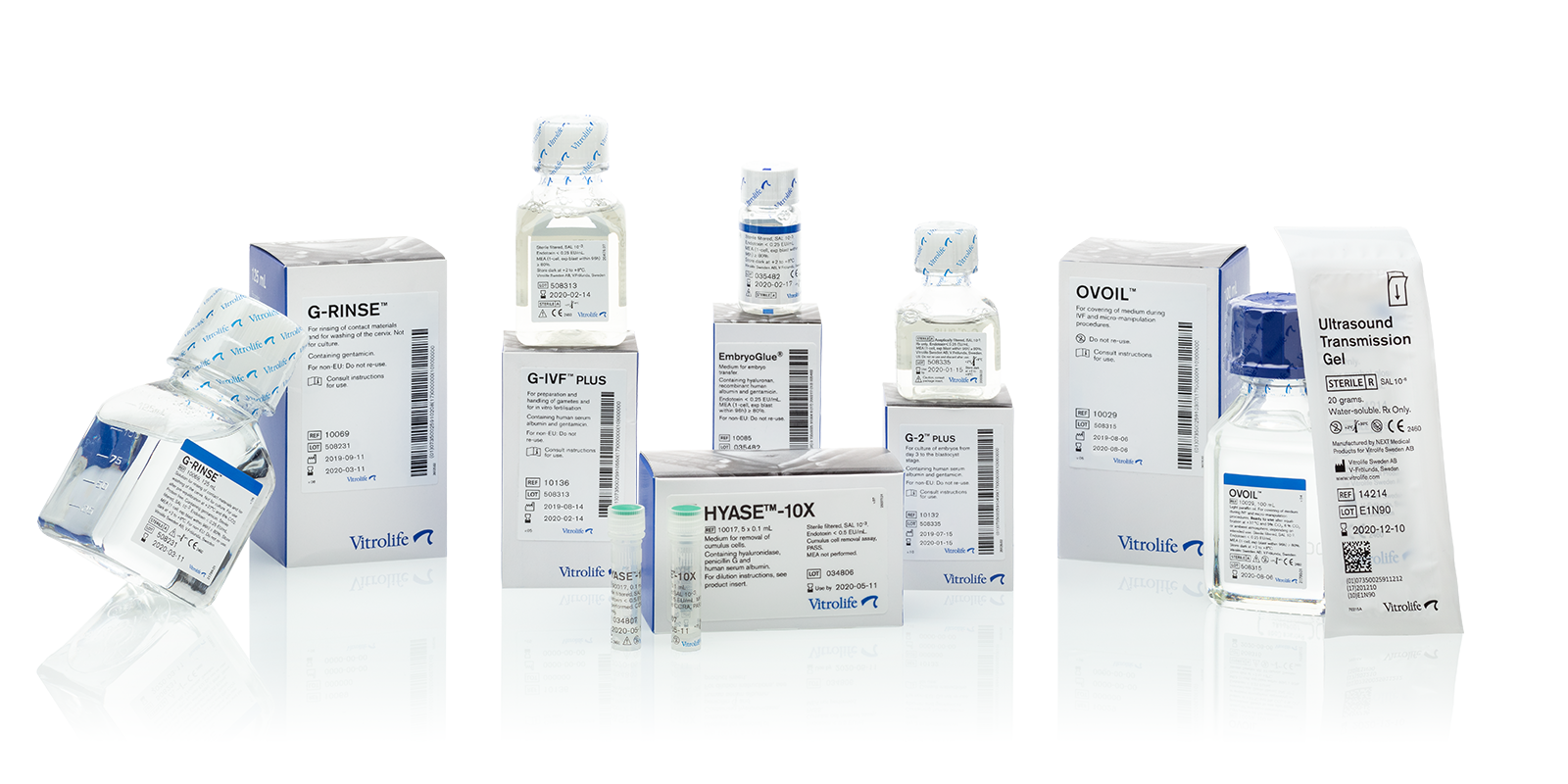
“A picture is worth a thousand words” – this old proverb is embraced and applied by medical device manufacturers all around the world, but why is that?
The answer to that question starts with the fact that most countries’ health authorities require medical device manufacturers to provide product information to the users in their local language. This is a not a problem for manufacturers who produce and distribute big volume products where an entire batch will be sold in only one country. They simply produce batches dedicated to the specific country with product information in that local language.
However, manufacturers who are producing specialised niche products which are being distributed globally face a much greater challenge. It is not possible to produce country specific batches since each batch will be distributed to perhaps 50 different countries. Instead the product information delivered with each product must be understandable globally, i.e. product information must include perhaps 30-50 different languages. This is not a problem when it comes to product inserts and manuals where you can use several pages or large folded sheets, but it is a problem when it comes to the actual label on the product, especially if the medical device is physically small.
Typical information required on the product label is:
- product name
- REF
- LOT
- intended use
- warnings
- sterilisation method
- expiry date
- manufacturing date
- manufactured by
- storage conditions
- reference to the product insert
- re-use information
- special product information
- bar code
Just the phrase “Manufactured by” would easily cover the entire surface of a label on a small medical device once translated into 50 different languages. This can be solved by using fold-out labels to increase the label area, but fold-out labels are often inappropriate to use in a clinical environment where all items need to be wiped down with alcohol to be aseptic or clean. Another solution is to make packages larger than the actual medical device itself to increase the area of the labels, but this is quite inconvenient in a hospital or clinic where storage space is limited and not good for the environment.
So, this is the reason medical device manufacturers embrace the pro-verb “A picture is worth a thousand words” – they print pictures on the labels to fit a thousand words in many different languages!
The pictures on the labels are made up of several symbols, or more correctly signs, as signs only have one meaning while symbols are often vaguer and more undefined. There are internationally recognized symbols which represent most of the different types of information above and they are listed in standard ISO 15223-1: 2016. By using these symbols, the manufacturer can fit more than a thousand words in all languages on a small label.
There is a condition for this to work, the health care professionals need to understand the symbols. If they do not understand the symbols this type of labelling will be useless of course. The most important symbol to understand is the symbol “Consult instructions for use”:
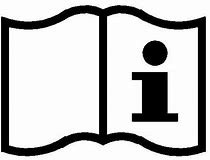
The meaning of each symbol should be explained with words in all relevant languages in the accompanying documentation such as the product insert or product manual. So, if the health care professionals at least understand the symbol “Consult instructions for use” the system will work and make sense.
There are more than 40 symbols in the standard which cover the most common information required for a medical device label. However, sometimes the labels need to include special information which are not recognized internationally. E.g. the symbol “Intended for removing cumulus cell complex in preparation for intra-cytoplasmatic sperm injection” cannot be found in the standard for symbols. The medical device manufacturer of IVF products can then design its own symbol for such specialized purposes. These must be explained together with the other symbols in the instructions for use as well.
So, what other symbols would you find on the labels of an IVF product?
As mentioned above the “Consult instructions for use” is important because it refers the user to the explanations of all the symbols and more information. Hence, if you do not understand that symbol you might not understand any other symbol unless you by chance read the instructions for use.
There are some self-explanatory symbols such as:
 followed by a number which means REF number (or catalogue number) and:
followed by a number which means REF number (or catalogue number) and:
 followed by a number which means LOT number (or batch number) – pretty intuitive to understand these two symbols!
followed by a number which means LOT number (or batch number) – pretty intuitive to understand these two symbols!
The symbol “Sterile” is also self-explanatory for the English speaking:
 However, the symbol “Sterile” can be followed by other symbols identifying the type of sterilisation method such as the symbol for aseptic processing which is the most common symbol for IVF media:
However, the symbol “Sterile” can be followed by other symbols identifying the type of sterilisation method such as the symbol for aseptic processing which is the most common symbol for IVF media:
 There are other sterilisation methods used for IVF instruments which are delivering a sterility assurance level of 10-6. Examples of those are sterilisation methods are sterilisation by dry heat or steam:
There are other sterilisation methods used for IVF instruments which are delivering a sterility assurance level of 10-6. Examples of those are sterilisation methods are sterilisation by dry heat or steam:
 or ethylene oxide sterilisation:
or ethylene oxide sterilisation:
 or irradiation:
or irradiation:

These added symbols are harder to interpret, but the main thing is usually that the device is sterile to avoid infections in the culture dish or patient. The way the product became sterile is not important as long as the manufacturer did not make the IVF product toxic or reduced the performance or safety of the device during the sterilisation process.
There are other symbols that are not so intuitive. The symbol “Manufacturer”:
 and the symbol “Date of manufacture”:
and the symbol “Date of manufacture”:

are similar and can easily be mixed up. Even if they are not so intuitive, you can distinguish them by the text or numbers that are adjacent to the symbol. “Manufacturer” is followed by the legal manufacturer name and address and is mandatory on IVF products. The “Date of manufacture” is followed by the manufacturing date and is usually seen on equipment which do not have an expiry date, but certain disposable IVF products with an expiry date will also be required to include the manufacturing date. These symbols can even be combined in one symbol according to the standard.
The symbol “Use-by date”:

followed by the expiry date is one of the most common symbols you can find on products, not just medical devices. Most people associates the hourglass with time that is running out, the product is expiring. This symbol is not at all intuitive since very few persons alive today have used an hourglass, but most people have been sitting for eons in front of a computer screen staring at the hourglass and cursing the latest software update. The symbol is mandatory on disposable IVF products, but not for equipment.
To understand under what conditions a product should be stored, the symbol “Temperature limit” is of course relevant:
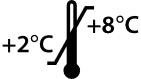
It is easy to understand for older users who actually have seen a mercury thermometer close up when they proposed to their parents to stay home from school due to fever, but younger generations might find it harder since thermometers used today are usually digital – just a display and a sensor and sometimes a cord. This symbol is always applicable for IVF media which usually requires an ambient temperature of +2 to +8 °C.
Finally, one symbol which is crucial for disposable IVF products is the symbol “Do not re-use”:
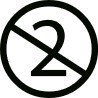
The symbol tells you that you may not re-use the product. It used to be common for clinics to re-use disposable devices which sometimes had a negative impact on the patient such as infections, cross-contamination between patients or reduced performance.
The Vitrolife media may be re-used if this symbol is missing. Just remember to work aseptically to avoid contamination and follow the instructions for use.
So, just the handful of symbols described above corresponds to more than a thousand words when translated to 50 languages. These symbols, and some specially designed symbols, are elements of the beautiful and important pictures you will find on the labels of IVF products, so much better than a thousand words in different languages. Think of this next time you look at a label of an IVF product and be grateful that you do not have to search for your specific language among 49 other languages to figure out the storage conditions of the product.
Topics: IVF community insights
Written by Hans Lehmann
As responsible manager for media development, regulatory affairs and post market, Hans has experience of the products and product labelling from the very initial development phase to the end of the product life cycle. Hans has been with Vitrolife for 20 years and has been involved numerous projects such as development of new products, global registrations, quality improvements and introduction of the environmental management system.