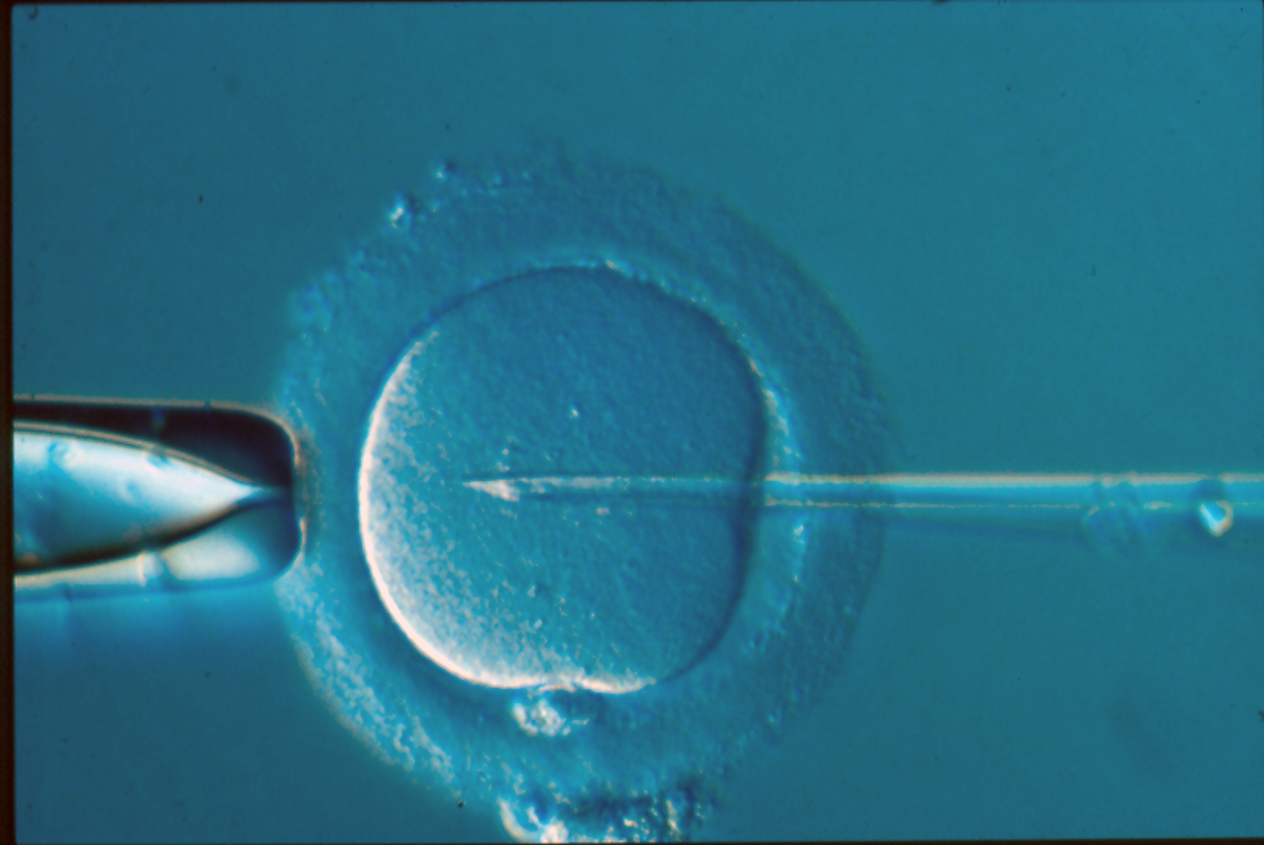
Different assisted fertilisation methods were developed and implemented in the field of IVF during 1980-1990. Out of these methods, intracytoplasmic sperm injection (ICSI) is the most successful method developed1 and is considered to be one of the major breakthroughs in this field since the birth of Louise Brown in 1978. While assisted fertilisation techniques, including ICSI, were introduced to overcome fertilisation failure in cases of severe male infertility, ICSI is today used in > 60 % of all IVF cycles globally.2
ICSI has frequently been described as 'an accident' in the lab. For me, being part of the development of the ICSI technique, I would rather describe ICSI as an inevitable consequence of the procedure used to perform subzonal insemination. The aim of this blog is, however, not to cover the history of ICSI but rather to focus on aspects that support successful ICSI in the IVF laboratory.
There are many challenges in the ICSI procedure. Besides a skilled embryologist the result is also dependent on the biological material and the tools used.
Protecting the oocyte
Maintaining physico-chemical conditions during procedures is of paramount importance.3 Unfortunately, people do - unintentionally - not always give this cell the attention it deserves. The following are some basic aspects to consider:
- Tubes in which follicular fluid is collected during follicle aspiration are not always continuously kept at 37°C.
- Avoid temperature drops in the medium in the dish. Effects of the temperature drops on oocyte spindle with potential irreversible effects have been well documented.4
- An oil overlay will help minimise temperature changes.
- Use media according to manufacturers’ recommendations. Not all media will give you a stable pH for a long enough period outside the CO2-incubator.
Ensure a protective environment during oocyte denudation
Firstly, it is paramount that oocytes are surrounded by cumulus cells in vitro. They provide a protective environment and allow further maturation of immature oocytes that are usually present in an oocyte cohort.
Secondly, it is crucial to maintain stable physio-chemical conditions. Performing procedures under oil helps provide a more stable environment.
Finally, for oocyte denudation the use of hyaluronidase should be considered. Today, the enzyme activity levels used are safe and with sufficient rinsing steps following exposure to the enzyme the oocyte is not negatively affected. This was not the case previously. In the early days of ICSI, hyaluronidase was used at higher levels. This affected laboratory results negatively.5
The performance of ICSI has a great impact
While ICSI is overall very successful, the way the injection procedure is performed can have a significant impact on the effectiveness of a treatment cycle. The ICSI procedure is another step where cells are exposed to conditions that can impact their developmental potential. Again, temperature, pH and osmolality are critical parameters. My colleague Jaco Terblanche has written an excellent blog about the importance of temperature measurements and temperature control. Read it here. Additionally, light exposure, operator skills as well as use of appropriate labware and microtools can also have an impact on results. You can download my white paper at the end of this blog for an elaborate discussion of these parameters.
Operators need sufficient training
ICSI is usually performed by multiple operators who may achieve different results. Sufficient training is thus important. The number of oocytes to be injected varies between operators. A statistical method to measure trainee performance shows that between 35 and 80 oocytes are needed for different trainees to reach the expected level of proficiency.6
Preparing for culture
During an ICSI procedure, oocytes may be moved between different types of media at different time points before culture starts. When changing between media with different types of buffers it is also important to properly rinse oocytes, minimising carry-over from a medium with different buffering capacities. Culture after ICSI is outside the scope of this blog, however avoiding unnecessary incubator door openings is a must to maintain stable and undisturbed culture conditions, and the optimal culture system is an integrated time-lapse incubator. Learn more in this blog post written by my colleague Dr. Markus Montag.
ICSI for all?
The current trend in IVF laboratories is one of optimisation and simplification. The increasing use of ICSI goes completely against this. ICSI is more labour intensive and exposes oocytes to many more additional stresses compared to oocytes where standard IVF is performed. The impact of these additional stresses can partly explain why ICSI is not superior when not indicated.7-9 So should we really perform ICSI for all patients?
Conclusion
While ICSI is a major step forward in human ART and allows couples to have their own genetic child if a single viable sperm cell for each oocyte can be injected, considering its current level of performance, its use should be questioned when not indicated. Besides, additional efforts are required to bring success rates for all clinics to the levels suggested by experts in the field. Still today, basic conditions such as temperature, osmolality and pH do not always get the necessary attention. Like for so many other things in the IVF lab, also for ICSI, the devil is in the details.
Download white paper to learn more
I have written a white paper where several important factors leading to successful ICSI will be discussed. Click to download the white paper. I hope you will enjoy the reading.
References:
1. Devroey P, Van Steirteghem A. A review of ten years experience of ICSI. Hum Reprod Update. 2004 Jan-Feb;10(1):19-28
2. Adamson GD, de Mouzon J, Chambers GM, Zegers-Hochschild F, Mansour R, Ishihara O, Banker M, Dyer S. International Committee for Monitoring Assisted Reproductive Technology: world report on assisted reproductive technology, 2011. Fertil Steril. 2018 Nov;110(6):1067-1080
3. Wale PL, Gardner DK. The effects of chemical and physical factors on mammalian embryo culture and their importance for the practice of assisted human reproduction. Hum Reprod Update. 2016 Jan-Feb;22(1):2-22
4. Almeida and Bolton, 1995; Wang et al., 2001; Gomes et al., 2012)
5. Joris H, Liu J, Nagy Z, Van Ranst H, Vankelcom A, Desmet B, Bocken G, Van Steirteghem AC. Influence of hyaluronidase concentration on the number of one-pronucleated oocytes after subzonal and intracytoplasmic sperm injection. Poster, Andrology in the nineties, Genk, Belgium 1993.
6. Durban et al. (2016),Durban M, García D, Obradors A, Vernaeve V, Vassena R. Are we ready to inject? Individualized LC-CUSUM training in ICSI. J Assist Reprod Genet. 2016 Aug;33(8):1009-15
7. Tannus S, Son WY, Gilman A, Younes G, Shavit T, Dahan MH. The role of intracytoplasmic sperm injection in non-male factor infertility in advanced maternal age. Hum Reprod. 2017 Jan;32(1):119-124
8. Sustar K, Rozen G, Agresta F, Polyakov A. Use of intracytoplasmic sperm injection (ICSI) in normospermic men may result in lower clinical pregnancy and live birth rates. Aust N Z J Obstet Gynaecol. 2019 Jun 11. doi: 10.1111/ajo.13004. [Epub ahead of print]
9. https://www.focusonreproduction.eu/article/ESHRE-Meetings-BOEA-2019-ICSI
Topics: Embryo culture & transfer
Written by Hubert Joris
Hubert is like a living library. What Hubert does not know about papers published in the field of IVF is not worth to know. Hubert is an experienced embryologist and was member of the Belgian group that invented the ICSI-method.