Mini-Series: How Terra Fertility is elevating care and supporting outcomes with EmbryoScope and PGT-A Plus
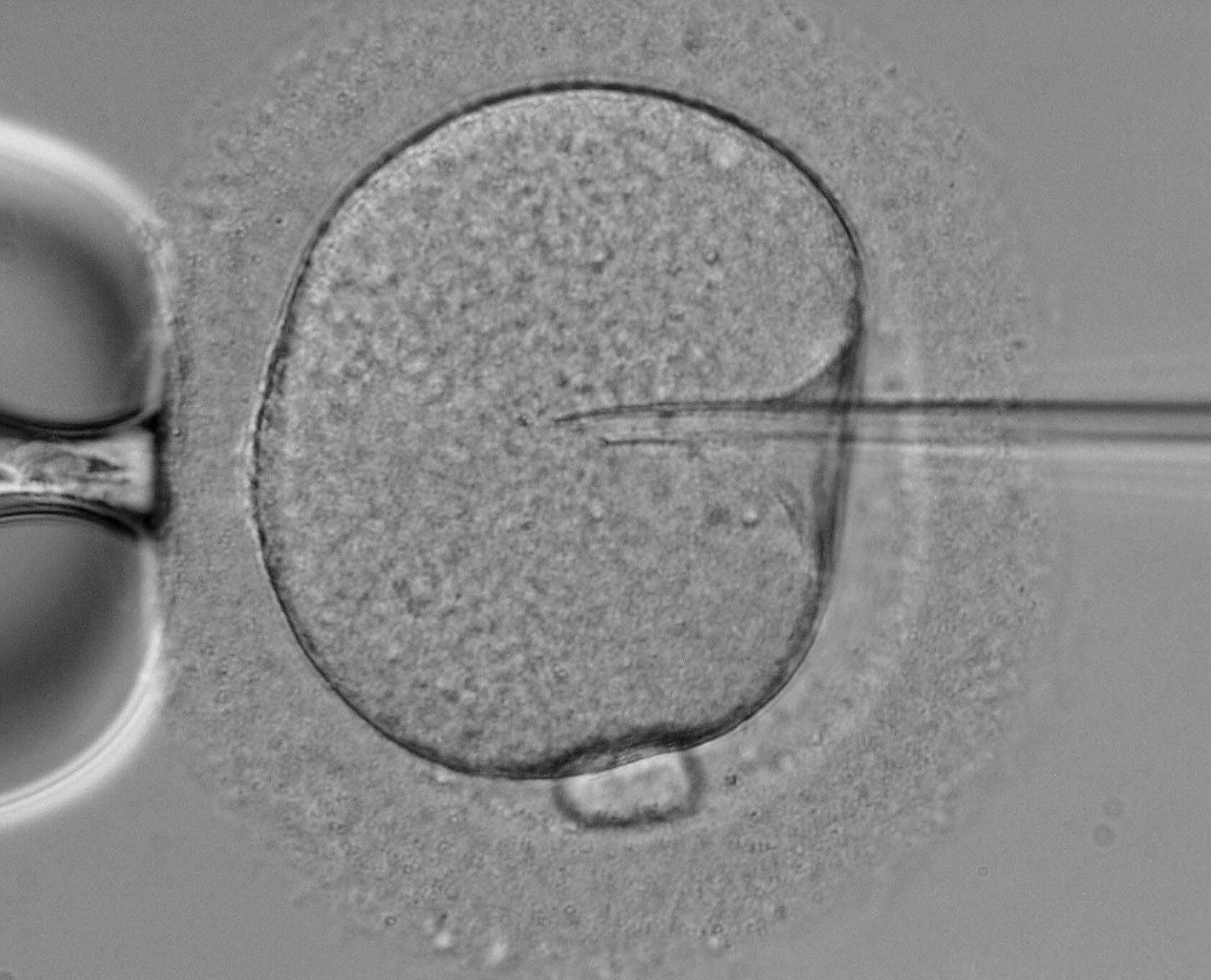
Data from a recently published clinical study showed a significant increase in detection of euploid/diploid embryos in embryos with confirmed atypical PN using time-lapse technology with EmbryoScope and ploidy detection.